People Living with Human Immunodeficiency Virus in Hadhramout: Clinical Presentation and Outcomes
Ahmed Mubarak Daakeek, Abdulrahim A. Bahashwan
Citation : Daakeek AM, Bahashwan AA. People Living with Human Immunodeficiency Virus in Hadhramout: Clinical Presentation and Outcomes. J Pathol Infect Dis 2019;2(1):1-9.
Objective: The objective of the study was to determine the clinical features and outcome of patients living with human
immunodeficiency virus (HIV) in Hadhramout and nearby governorates.
Materials and Methods: This descriptive study was conducted in the antiretroviral therapy (ART) site at Ibn-Sina General Hospital, Mukalla, Hadhramout governorate. All 145 patients were enrolled in HIV treatment and care program from December 2008 to the end of December 2016 with confirmed HIV test. Data included all personal data, clinical staging, drugs taken, and outcomes. Patients were grouped according to the decades to five groups, =15 years, 16-30 years, 31-50 years, 51-70 years, and >70 years. Cases classify according to the antiretroviral drugs to ART group and Pre-ART group. The relevant data parameters were analyzed using SPSS statistical software version 21 and Excel 10.
Results: A total of 145 cases, most adults (97.9%), males and females were104 (71.7%) and 41 (28.3%), respectively. Mean age was 36.46 years and 30-50 years the most affected age group (55.2%). Clinical Stages 3 and 4 were the common presentation in 73.8%, and most cases were from Mukalla city. Of the total cases, 74.5% were on ART 53.1 of them improved, pulmonary tuberculosis was found in 4 cases, and death cases were (18.5%), mostly due to late presentation and non-adherence, and mostly occurred in early 6 months of starting the ART. 37 patients were in a pre-treatment group (21.6%), where the mortality rate is 35.1%, mainly due to loss of follow-up.
Conclusions: Most cases were adult males, young age and have had late presentation, where mortality is higher in the pre-treatment group due to loss of follow-up and in early 6 months of treatment.
Keywords: Antiretroviral therapy, deaths, Hadhramout, lost to follow-up, Pre-antiretroviral therapy
INTRODUCTION
The acquired immunodeficiency syndrome (AIDS) was first recognized in 1981, although the earliest documented case has been traced to a blood sample from 1959. It is caused by the human immunodeficiency virus (HIV-1), which is a member of the Retroviridae family that replicates in CD4 positive cells, which has evolved a number of mechanisms to elude immune control and has thereby prevented effective control of the epidemic [1]. HIV-2 causes a similar illness to HIV-1 but is less aggressive and restricted mainly to Western Africa [1].
AIDS remains the second leading cause of disease burden worldwide although downward trends in prevalence are occurring in several countries, partly as a result of effective prevention measures and partly due to scaling up of anti-retroviral access [1]. The center for disease control reported a significant increase in morbidity and mortality between 2001 and 2012 in the Middle East and North Africa region [2]. Highly active antiretroviral therapy (ART) with three or more drugs has improved life expectancy to near normal in the majority of patients with an 80% reduction of mortality[1].
Early identification of infected persons helps in reducing transmission, preventing complications, and modifying risk behavior [3]. Early diagnosis is important for appropriate and effective ART. The number of CD4+T lymphocytes at the time of confirming HIV-1 infection (CD4 - HIV) has been suggested to provide useful staging information [3]. The stage of disease at diagnosis is influenced by patients' willingness to be tested, their knowledge of the disease, and the effectiveness of the health-care system in finding and contact tracing [3].
Yemen is one of the countries with low prevalence of HIV (0.2%) in the general population, according to 2011 HIV size estimates by national AIDS program (NAP) [4]. In 2009, the estimated number of people living with HIV (PLHIV) was 23,000, and the cumulative number of HIV reported cases (since the first case in 1987) to 2009 is 2,882 [5]. There are no available data about the cumulative number of HIV related deaths and orphans. The main mode of HIV transmission was unprotected heterosexual contact 62%, homosexual 7%, and through mother to child transmission 5% and about 24% of the reported cases did not have a mode of transmission reported [5].
In Yemen, there are five sites providing services for PLHIV called ART sites, one of them was at Ibn-Sina general hospital in Al-Mukalla city, capital of Hadhramout governorate, which cover the four governorates of Hadhramout sector (Hadramout, Shabwa, Al-Mahra, and Socotra). This site is providing voluntary counseling and testing, post-exposure prophylaxis, prophylaxis for opportunistic infections and ART.
This study will highlight the general features, adherence, and outcome for PLHIV in ART site at Ibn-Sina hospital.
MATERIALS AND METHODS
This descriptive study was conducted at ART site in Ibn-Sina General Hospital for all PLHIV who are enrolled in HIV treatment and care since the start of the program from December 2008 to the end of December 2016.
A total of 145 cases registered including those who were transferred from other sites in the republic. A data collected from electronic records (Tier.net) including age, gender, residency, month of diagnosis, clinical stages on presentation, date of starting ART, and laboratory data. The age further subdivided based on decades into five groups; =15 years, 16-30 years, 31-50 years, 51-70 years, and >70 years.
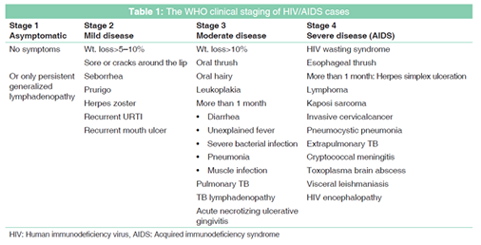
Clinical stages according to the WHO staging [6] was used in all cases [Table 1]. All PLHIV were defined as: (i) Pre-ART group included cases on prophylaxis treatment and not started treatment with ART, and (ii) ART group including cases treated with ART drugs and prophylaxis treatment. Treatment with ART was based on the WHO clinical staging and National AIDS program (NAP) policy which includes seven requirements mentioned in Table 2.

Lost to follow-up (LTFU) which defined as loss of follow-up for more than 6 months in pre-ART group and not attended three scheduled visits to the site in case of ART group. Dead cases were documented and the duration from diagnosis and started ART and death or loss to follow-up were registered.
Laboratory confirmation of HIV infection was based on detection of antibodies by ELISA, and two tested were done, one in hospital lab and another in a central lab. CD4 was not done for most of the cases due to unavailability in our hospital.
Data were analyzed using Microsoft Excel 2010 and SPSS statistical software version 21. The frequencies of different categorical variables were calculated. The median and range were calculated for continuous variables. Patient characteristics were compared using the Chi-square test or Fisher's exact test for categorical variables.
RESULTS
Overview of the Problem
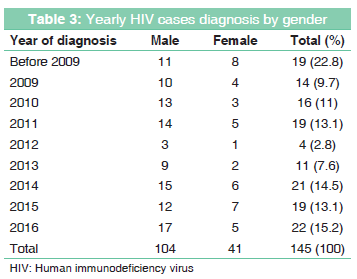
A total number of 145 PLHIV registered in the ART site during an 8 years period. Yearly diagnosed cases by gender are shown in Table 3. The cases increased gradually each year and the past 3 years (2014-2016) showed the highest number of cases had been registered. 33 death cases were reported resulting in a mortality rate of approximately (22.8%) among all documented cases.
Demographics Features
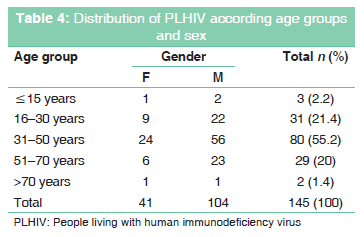
From all cases reported; 104 (71.7%) were male and 41 (28.3%) were females with male to female ratio of 2.5:1. The mean age when enrolled in ART site was 36.46 years, and the median was 35 years (range 3-83 years). Women were similar to men at diagnosis, mean of 36.12 (standard deviation [SD] ± 15.37) years versus 36.6 (SD ± 11.97) years for men, P = 4.88. Most of the cases were adults (97.9%), the most affected age group was from 30 to 50 years (55.2%), and their only 3 cases (2.2%) of children equal or below 15 years of age [Table 4]. One death case was reported in children < 15 years of age.
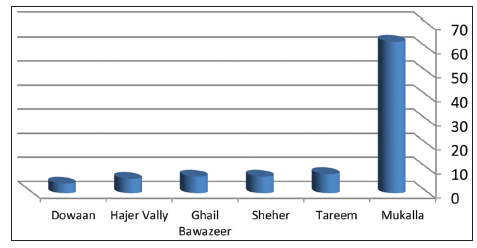
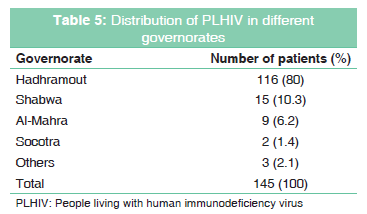
Patients belonging to Hadhramout governorate outnumbered and were 116 (80.0%) followed by 15 patients (10.3%) belonging to Shabwa governorate and 9 patients (6.2%) belong to Al-Mahra governorate. Representatives from other governorates were minimal (3.5%) [Table 5]. Mukalla city, capital of Hadhramout had the largest share of cases compared to other areas of the governorate (n = 64, 54.7%). Distribution of cases in common affected districts in Hadramout governorate [Figure 1].
Clinical Features
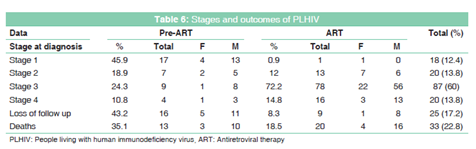
The most common presentation at the time of diagnosis was Stage 3 (n = 87.60%) followed by Stages 2 and 4 equally represented (n = 20, 13.8%) and Stage 1 (n = 18,12.4) [Table 6]. The CD4 was done in 15 cases in other governorates, and it was < 350 in 13 cases. 4 cases had pulmonary tuberculosis comorbidity. 2 cases were pregnant on diagnosis.
Classification According to Treatment
Pre-ART group: PLHIV not taken ART called pre-ART group and it was 37 cases (21.6% of all cases), 29 males (78.4%) and 8 females (21.6%). Most of this pre-ART group were present in clinical stage 1 and 2 (64.9%)[ Table 6], and their ages at presentation were between 30-50 years old.
ART group: PLHIV who undergone ART were placed in ART group. 108 cases were included in this group approximately (74.5%) of all cases. 75 cases were males (69.4%) and 33 (30.6%) were females. Two of female cases were pregnant. 4 cases had pulmonary tuberculosis. 50% of cases in the ART group belong to the 30-50 years age group. Clinical staging of this group at presentation in order of frequency; Stage 3 (n = 78, 72.2%), Stage 4 (16, 14.8%), Stage 2 (n = 13.12%), and final Stage 1 (n = 1, 0.9%) [Table 6]. Most cases started on zidovudine, lamivudine, and nevirapine regimen (46.3%) and tenofovir, emtricitabine, and efavirenz (38.9%).
Outcome
Most cases on ART improved symptomatically, with 77, 53.1% switching to Stage 1 and 6, 4.6% to Stage 2, and 5, 3.4% remaining on Stage 3 and no cases in Stage 4. For all enrolled cases in the program (pre-ART and ART groups), 25 cases were LTFU and this represented 17.2% of all cases (16 cases were from pre-ART groups and 9 cases from ART group), while 33 (22.8%) were died.
Deaths Cases
From all death cases, 26 cases (78.8%) males and 7 cases females (21.2%) with male to female ratio 3.7:1. Most of the death cases (75.8%) were on Stage 3 and 4. The duration from diagnosis to death was ranging from days to 89 months, with mean 22.25 (SD + 25.335). There is a significant correlation between duration of disease and deaths (P = 0.00) indicating most cases died in earlier 2 years of disease.
Pre-ART group deaths were 13 cases, 10 (76.9%) males and 3 (23.1%) females, with male to female ratio 3.3:1. Most cases in 31-50 years age group (76.9%) [Figure 2] with a mean (44 ± SD 7.439 years). Most death cases were either loss of follow-up (84.6%) or refuse treatment. Duration from diagnosis to death range from 0 to 62 months with a mean (15.23 ± SD 18.426); with most cases died in the first 2 years from diagnosis (84.6%) with correlation from time of diagnosis and deaths (P = 0.000).
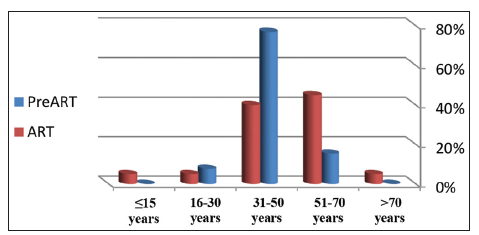
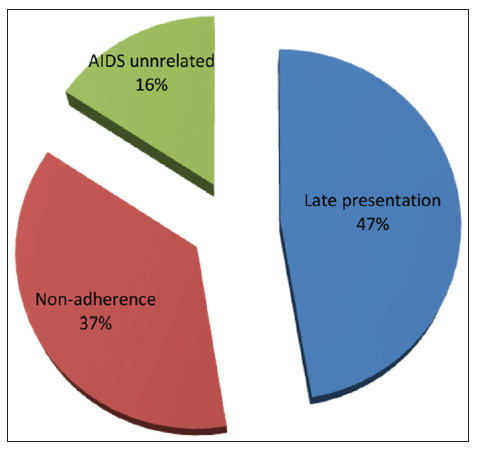
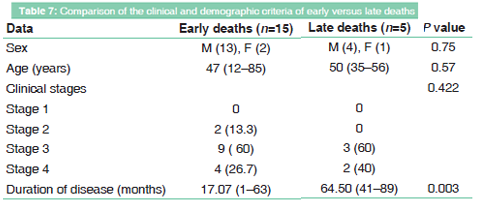
Death from ART group outnumbered 20 cases (18.5%), most of death cases were males (n = 16, 80%) [Table 7] with male to female ratio 4:1. Most cases in age range between 31 and 70 years (95%) [Figure 2] with mean (47.45 ± SD 15.500 years). Factors predisposing to death (based on clinical follow-up of ART team) vary from late stages of starting ART (n = 9.47%), non-adherence (n = 9.37%) which may relate to recurrent LTFU (6 cases), severe depression (3cases), and AID-unrelated causes such as trauma, postsurgical, and sudden death (n = 3.16%) [Figure 3].
Duration from starting ART to dead range from 1 month to 89 months with mean (27.05 ± SD 28.621); and most cases died within < 6 months from starting treatment (n = 16.80%) indicating a correlation between adherence to therapy for prolonged period and good response (P = 0.003) [Table 7].
DISCUSSION
The prevalence of HIV infection among people in the Middle East is relatively lower than other developing countries. However, Yemen, Saudi Arabia, Lebanon, and Libya the incidents of HIV infection were < 10% [7].
This is a retrospective study of HIV patients at an ART site in Ibn-Sina Hospital, Hadhramout Yemen, over the past 8 years. This site is being a main referral center for HIV infection in Hadhramout district which includes four governorates.
In our study, there were annually increases in the diagnosis of HIV cases which enrolled in ART services and this also seen in Saudi Arabia which is one of the low prevalence countries [8]. The majority of cases were males (71.7%) with male:female ratio 2.5:1 and this similar to other studies in Saudi Arabia [3,9] and higher than other study among Saudi patient where the ratio (4.4:1) [8]. The mean and median age of cases was 36.46 and 35 years, which somewhat higher than other studies were done in Saudi Arabia,[3,10] and similar to one Austrian study[11] with no differences between male and females mean and median ages differ than what seen in Saudi Arabia [3,10] where female younger than males. Most cases were adult 97.9% higher than that seen in one Saudi study where adult percent was (80%) in Saudi cases [8].
Most of our cases were from Hadhramout governorate and its main towns, especially Mukalla city and this perhaps may due to the existence of HIV treatment and care services in Mukalla city - Hadhramout governorate. On the other hand, AIDS-related stigma plays a major role in the access of a patient to services in whole Yemen[4], especially in rural areas. Hadhramout population has somewhat higher education level in comparing to other governorates, and the population of Al-Mahra governorate population mostly treated in neighbor countries especially Oman.
This study showed late stages were the presentation of most cases who enrolled in the ART program (approximately 73.8%) which considered AIDS-defining diseases. This is similar to one study done in Oman [12] and differs from studies in Saudi Arabia where the majority was asymptomatic at diagnosis [13-15].
The tuberculosis - HIV coinfection was a well-known entity in different literatures,[16-18] and tuberculosis cases were increases in an era of HIV/AIDS. In our study, there are 4 cases (2.76%) proved has pulmonary tuberculosis out of all cases enrolled in the program. This is in contrast to studies done in Jeddah in Saudi Arabia [19] and China [20] where the majority of cases has tuberculosis.
Most of PLHIV were under antiretroviral drugs (ART group) (74.6%), while the remaining cases are still in pre-ART group for three different reasons, first: They were in clinical Stages 1 and 2 which were not eligible to start treatment, according to the previous WHO recommendation, [6] second: Loss of follow-up which constitute about 43.2% and this possibly due to fear of stigma and discrimination, cost of travelling to site for cases from distant places, low education level and seeking a traditional treatment elsewhere, and third: Feeling healthy and refuse to take the ART. This is also seen in different studies in Asia and Africa [21-25].
From this pre-ART group most cases where either died (35.1%) or LTFU (43.2%) which constitute (78.3%) of all cases, and the remaining cases (8 cases, 21.7%) still not started ART drugs, and for that reasons the outcome was very poor as the studies reported the benefits of early therapy and bad effect of LTFU on outcomes [23,26,27].
The ART group which considered the main group in the site (74.5), the late presentation (Stages 3 and 4) was common in approximately 87%, the most common affected age group was 30-50 years and male were predominant. The drug regimens used were zidovudine, lamivudine, and nevirapine regimen in (46.3%) and tenofovir, emtricitabine, and efavirenzin (38.9%) which recommended by the WHO and NAP and used in different countries [14] and improved efficacy in one study done in Sana'a-Yemen [28].
There is a favorable clinical outcome in the ART group, with improvement in about 57.7% of cases, and these were mentioned in literature,[29,30] and a major obstacle in this group was the LTFU which constitute about 8.3% of total cases. The causes are the same as mentioned earlier in the pre-ART group, and this problem was seen in different studies, especially in African countries [25,27,31].
Death cases were about 22.8%, and this was similar to a neighboring country (Saudi Arabia)[10]. Most of the death cases in both groups were found in early 2 years after diagnosis indicating a strong correlation between risk of death and time after diagnosis, and late presentation as one of the major causes of death [32].
The death cases in pre-ART group about 35.8% of all cases in the group, most of them were male in the age group 51-70 years, the majority of them died during the period of LTFU (84.6), and the others due to late presentation.
Mortality in ART group was about 18.5% of total cases, and most of the cases were male, and this rate is very high in the context of a patient taking ART drugs and higher than African countries [33]. This attributed to different factors leading to deaths and the most important causes, according to the clinical judgments were: (a) Late presentation of a large group of patient and this is documented problem leading to death,[32] (b) non-adherence to treatment, and this may be due to recurrent LTFU due to effect of depression, stigma, and discrimination, poverty and difficult to reach to the site, beliefs about the necessity of ART, low educational level, and defect in counseling before starting treatment. All these factors are found as barriers to adherence in multiple studies done elsewhere in the world [34-42].
In our study, we found that the majority of death cases in ART group was dying in the first 6 months of starting ART drugs (P = 0.003), and the outcome was improved with better adherence and close follow-up, thereafter, indicating longer time spent receiving ART associated with improved survival and deaths mostly from non-AIDs related causes; this finding is also documented in different studies that outcome is sometimes unpredicted in the early period of treatment from 3 months to 3 years mostly in low-income countries in which our country is one of them [43-48].
CONCLUSIONS
Our study demonstrated that most PLHIV were younger age, mostly males, presented mostly in late stages, with a significant proportion of them LTFU and death. Death cases were improved with prolonged taking ART and with early starting it in asymptomatic patients, and higher efficacy of first-line triple therapy in a higher proportion of cases.
The limitations of our study were mostly unavailability of immunological and virological measures in the form of CD4 and polymerase chain reaction studies for evaluation of cases, staging, and follow their managements accurately.
ACKNOWLEDGMENTS
We would like to acknowledge ART staff, especially Dr. Nasreen Daakeek for her assistance with the correction of data, and Mr. Abdulla Bazarah and Mr. Fahed Bajar fan who supported and facilitating our job.
REFERENCES
- Walker BR, Colledge NR. Davidson's Principles and Practice of Medicine. 21st ed. Ch. 14. Amsterdam: Elsevier Health Sciences; 2013. p. 1-2.
- Ghasemian A, Mostafavi SK, Vardanjani AR. Human immunodeficiency virus (HIV) distribution in Middle East region. Avicenna J Clin Microbiol Infect 2016;3. Available from: http://www.ajcmicrob.com/?page=article&article_id=35965. [Last cited on 2017 Mar 04].
- Alrajhi AA, Halim MA, Al-Abdely HM. Presentation and reasons for HIV-1 testing in Saudi Arabia. Int J STD AIDS 2006;17:806-9.
- Ghouth AS. Role of stigma and discrimination in people living with HIV (PLHIV) in Hadramout governorate/Yemen. J Community Med Public Health Care 2016;3:11.
- Mohammed MF. Access to voluntary counseling and testing services for female sex workers and men who have sex with men in Yemen. J Infect Dis The 2016;4:264.
- World Health Organization. Chronic HIV Care with ARV Therapy and Prevention. IMAI/IMCI. Geneva: World Health Organization; 2007.
- Abu-Raddad LJ, Hilmi N, Mumtaz G, Benkirane M, Akala FA, Riedner G, et al. Epidemiology of HIV infection in the middle East and North Africa. AIDS 2010;24 Suppl 2:S5-23.
- Mazroa MA, Kabbash IA, Felemban SM, Stephens GM, Al-Hakeem RF, Zumla AI, et al. HIV case notification rates in the kingdom of Saudi Arabia over the past decade (2000-2009).PLoS One 2012;7:e45919.
- Alrajhi AA, Halim MA, Al-Abdely HM. Mode of transmission of HIV-1 in Saudi Arabia. AIDS 2004;18:1478-80.
- Al-Mozaini MA, Mansour MK, Al-Hokail AA, Mohmed MA, Daham MA, Al-Abdely HM, et al. HIV-care outcome in Saudi Arabia; A longitudinal cohort. J AIDS Clin Res 2014;5:370.
- Correll PK, Law MG, McDonald AM, Cooper DA, Kaldor JM. HIV disease progression in Australia in the time of combination antiretroviral therapies. Med J Aust 1998;169:469-72.
- Balkhair AA, Al-Muharrmi ZK, Ganguly S, Al-Jabri AA. Spectrum of AIDS defining opportunistic infections in a series of 77 hospitalised HIV-infected Omani patients. Sultan Qaboos Univ Med J 2012;12:442-8.
- Memish ZA, Al-Tawfiq JA, Filemban SM, Qutb S, Fodail A, Ali B, et al. Antiretroviral therapy, CD4, viral load, and disease stage in HIV patients in Saudi Arabia: A 2001-2013 cross-sectional study. J Infect Dev Ctries 2015;9:765-9.
- Isaac A, Ali B, Qutb S, Flemban S. Antiretroviral therapy,CD4, viral load, and disease stage in Saudi HIV patients: A cross sectional study of cases diagnosed between 2001-2013.J AIDS Clin Res 2015;6:447.
- Madani TA, Al-Mazrou YY, Al-Jeffri MH, Al Huzaim NS. Epidemiology of the human immunodeficiency virus in Saudi Arabia; 18-year surveillance results and prevention from an Islamic perspective. BMC Infect Dis 2004;4:25.
- Corbett EL, Watt CJ, Walker N, Maher D, Williams BG, Raviglione MC, et al. The growing burden of tuberculosis: Global trends and interactions with the HIV epidemic. Arch Intern Med 2003;163:1009-21.
- Raviglione MC, Harries AD, Msiska R, Wilkinson D, Nunn P.Tuberculosis and HIV: Current status in Africa. AIDS 1997;11 Suppl B: S115-23.
- Bevilacqua S, Rabaud C, May T. HIV-tuberculosis coinfection. Ann Med Interne (Paris) 2002;153:113-8.
- Sharton AM, Alturkistani EL, Alayyafi WA, Alnafisah HA, Khan AH, Alzein EH, et al. Prevalence of tuberculosis among HIV patients in Jeddah, Saudi Arabia. Egypt J Hosp Med 2017;66:257-60.
- Jiang X, Lu H, Zhang Y, Zhou Z, Ye H, Zhao Q, et al. A cross-sectional study of HIV and tuberculosis coinfection cases in Mainland China. South Med J 2008;101:914-7.
- Kalichman SC, Catz S, Ramachandran B. Barriers to HIV/ AIDS treatment and treatment adherence among African-American adults with disadvantaged education. J Natl Med Assoc 1999;91:439-46.
- Rangarajan S, Tram HN, Todd CS, Thinh T, Hung V, Hieu PT, et al. Risk factors for delayed entrance into care after diagnosis among patients with late-stage HIV disease in Southern Vietnam. PLoS One 2014;9:e108939.
- Geng EH, Bwana MB, Muyindike W, Glidden DV, Bangsberg DR, Neilands TB, et al. Failure to initiate antiretroviral therapy, loss to follow-up and mortality among HIV-infected patients during the pre-ART period in Uganda.J Acquir Immune Defic Syndr 2013;63:e64-71.
- Losina E, Bassett IV, Giddy J, Chetty S, Regan S, Walensky RP, et al. The "ART" of linkage: Pre-treatment loss to care after HIV diagnosis at two PEPFAR sites in Durban, South Africa. PLoS One 2010;5:e9538.
- Micek MA, Gimbel-Sherr K, Baptista AJ, Matediana E, Montoya P, Pfeiffer J, et al. Loss to follow-up of adults in public HIV care systems in central Mozambique: Identifying obstacles to treatment. J Acquir Immune Defic Syndr 2009;52:397-405.
- Grinsztejn B, Hosseinipour MC, Ribaudo HJ, Swindells S, Eron J, Chen YQ, et al. Effects of early versus delayed initiation of antiretroviral treatment on clinical outcomes of HIV-1 infection: Results from the phase 3 HPTN 052 randomised controlled trial. Lancet Infect Dis 2014;14:281-90.
- Rachlis B, Ochieng D, Geng E, Rotich E, Ochieng V,Maritim B, et al. Implementation and operational research: Evaluating outcomes of patients lost to follow-up in a large comprehensive care treatment program in Western Kenya. J Acquir Immune Defic Syndr 2015;68:e46-55.
- Doa'a AI, Elnemr MA. Assessment of the effectiveness of two strategies of antiretroviral therapy in Yemen. Yemeni J Med Sci2010;4:7.
- Hogg RS, Yip B, Kully C, Craib KJ, O'Shaughnessy MV,Schechter MT, et al. Improved survival among HIV-infected patients after initiation of triple-drug antiretroviral regimens. CMAJ 1999;160:659-65.
- Improved Survival Among HIV-Infected Individuals Following Initiation of Antiretroviral Therapy|HIV|JAMA|The JAMA Network. Available from: http://www.jamanetwork.com/ journals/jama/fullarticle/187227. [Last cited on 2017 Mar 04].
- Mugisha V, Teasdale CA, Wang C, Lahuerta M, Nuwagaba-Biribonwoha H, Tayebwa E, et al. Determinants of mortality and loss to follow-up among adults enrolled in HIV care services in Rwanda. PLoS One 2014;9:e85774.
- Sobrino-Vegas P, Moreno S, Rubio R, Viciana P, Bernardino JI, Blanco JR, et al. Impact of late presentation of HIV infection on short-,mid-and long-term mortality and causes of death in a multicenter national cohort: 2004-2013. J Infect 2016;72:587-96.
- Geng EH, Odeny TA, Lyamuya RE, Nakiwogga-Muwanga A,Diero L, Bwana M, et al. Estimation of mortality among HIV-infected people on antiretroviral treatment in East Africa: A sampling based approach in an observational, multisite, cohort study. Lancet HIV 2015;2:e107-16.
- Langebeek N, Gisolf EH, Reiss P, Vervoort SC, Hafsteinsdottir TB, Richter C, et al. Predictors and correlates of adherence to combination antiretroviral therapy (ART) for chronic HIV infection: A meta-analysis. BMC Med 2014;12:142.
- Catz SL, Kelly JA, Bogart LM, Benotsch EG, McAuliffe TL. Patterns, correlates, and barriers to medication adherence among persons prescribed new treatments for HIV disease. Health Psychol 2000;19:124-33.
- Cornell M, Lessells R, Fox MP, Garone DB, Giddy J, Fenner L, et al. Mortality among adults transferred and lost to follow-up from antiretroviral therapy programmes in South Africa: A multicenter cohort study. J Acquir Immune Defic Syndr 2014;67:e67-75.
- Lyimo RA, Stutterheim SE, Hospers HJ, de Glee T, van der Ven A, de Bruin M, et al. Stigma, disclosure, coping, and medication adherence among people living with HIV/ AIDS in Northern Tanzania. AIDS Patient Care STDS 2014;28:98-105.
- Sayles JN, Wong MD, Kinsler JJ, Martins D, Cunningham WE. The association of stigma with self-reported access to medical care and antiretroviral therapy adherence in persons living with HIV/AIDS. J Gen Intern Med 2009;24:1101-8.
- Katz IT, Ryu AE, Onuegbu AG, Psaros C, Weiser SD, Bangsberg DR, et al. Impact of HIV-related stigma on treatment adherence: Systematic review and meta-synthesis. J Int AIDS Soc 2013;16:18640.
- Abaasa AM, Todd J, Ekoru K, Kalyango JN, Levin J, Odeke E, et al. Good adherence to HAART and improved survival in a community HIV/AIDS treatment and care programme: The experience of the AIDS support organization (TASO),Kampala, Uganda. BMC Health Serv Res 2008;8:241.
- HIV TV, Counseling, Group TES, others. Efficacy of voluntary HIV-1 counselling and testing in individuals and couples in Kenya, Tanzania, and Trinidad: A randomised trial. The voluntary HIV-1 counseling and testing efficacy study group. Lancet 2000;356:103-12.
- Glass TR, De Geest S, Weber R, Vernazza PL, Rickenbach M, Furrer H, et al. Correlates of self-reported nonadherence to antiretroviral therapy in HIV-infected patients: The Swiss HIV cohort study. J Acquir Immune Defic Syndr 2006;41:385-92.
- Braitstein P, Brinkhof MW, Dabis F, Schechter M, Boulle A, Miotti P, et al. Mortality of HIV-1-infected patients in the first year of antiretroviral therapy: Comparison between low-income and high-income countries. Lancet 2006;367:817-24.
- Grabar S, Le Moing V, Goujard C, Leport C, Kazatchkine MD, Costagliola D, et al. Clinical outcome of patients with HIV-1 infection according to immunologic and virologic response after 6 months of highly active antiretroviral therapy. Ann Intern Med 2000;133:401-10.
- Ledergerber B, Egger M, Opravil M, Telenti A, Hirschel B, Battegay M, et al. Clinical progression and virological failure on highly active antiretroviral therapy in HIV-1 patients: A prospective cohort study. Swiss HIV cohort study. Lancet 1999;353:863-8.
- Ingle SM, May MT, Gill MJ, Mugavero MJ, Lewden C, Abgrall S, et al. Impact of risk factors for specific causes of death in the first and subsequent years of antiretroviral therapy among HIV-infected patients. Clin Infect Dis 2014;59:287-97.
- Brennan AT, Long L, Useem J, Garrison L, Fox MP. Mortality in the first 3 months on antiretroviral therapy among HIV-positive adults in low-and middle-income countries: A meta-analysis. J Acquir Immune Defic Syndr 2016;73:1-10.
- Palella FJ Jr., Baker RK, Moorman AC, Chmiel JS, Wood KC, Brooks JT, et al. Mortality in the highly active antiretroviral therapy era: Changing causes of death and disease in the HIV outpatient study. J Acquir Immune Defic Syndr 2006;43:27-34